
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB4 4 tetrahedral tetrahedral trigonal pyramidal AB3E 3 1 tetrahedral VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry trigonal planar trigonal planar AB3 3 trigonal planar AB2E 2 1 bent VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB2 2 linear linear AB3 3 trigonal planar AB4 4 tetrahedral tetrahedral AB5 5 trigonal bipyramidal AB6 6 octahedral octahedralġ3 > bonding-pair vs. VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB2 2 linear linear AB3 3 trigonal planar AB4 4 tetrahedral tetrahedral trigonal bipyramidal trigonal bipyramidal AB5 5 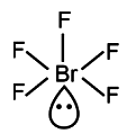
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB2 2 linear linear AB3 3 trigonal planar AB4 4 tetrahedral tetrahedral VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB2 2 linear linear trigonal planar trigonal planar AB3 3 Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB2 2 linear linear B Predict the geometry of the molecule from the electrostatic repulsions between the electron (bonding and nonbonding) pairs. Permission required for reproduction or display.Ģ Valence shell electron pair repulsion (VSEPR) model: Permission required."- Presentation transcript:ġ Chemical Bonding II: Molecular Geometry and Hybridization of Atomic OrbitalsĬhapter 10 Copyright © The McGraw-Hill Companies, Inc. Presentation on theme: "Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals Chapter 10 Copyright © The McGraw-Hill Companies, Inc.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
